Research on chemical modification of nickel coating on diamond particles surface
-
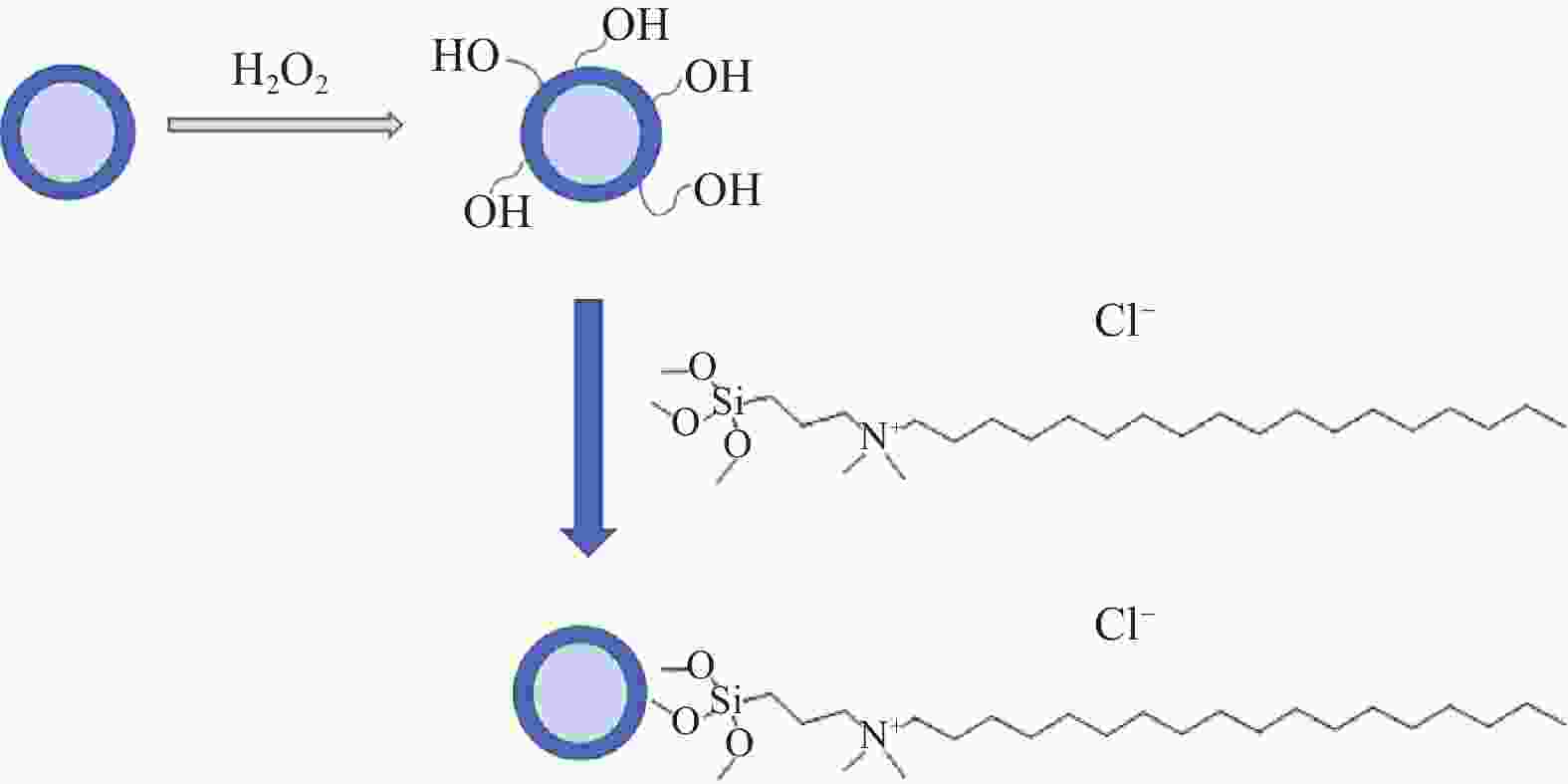
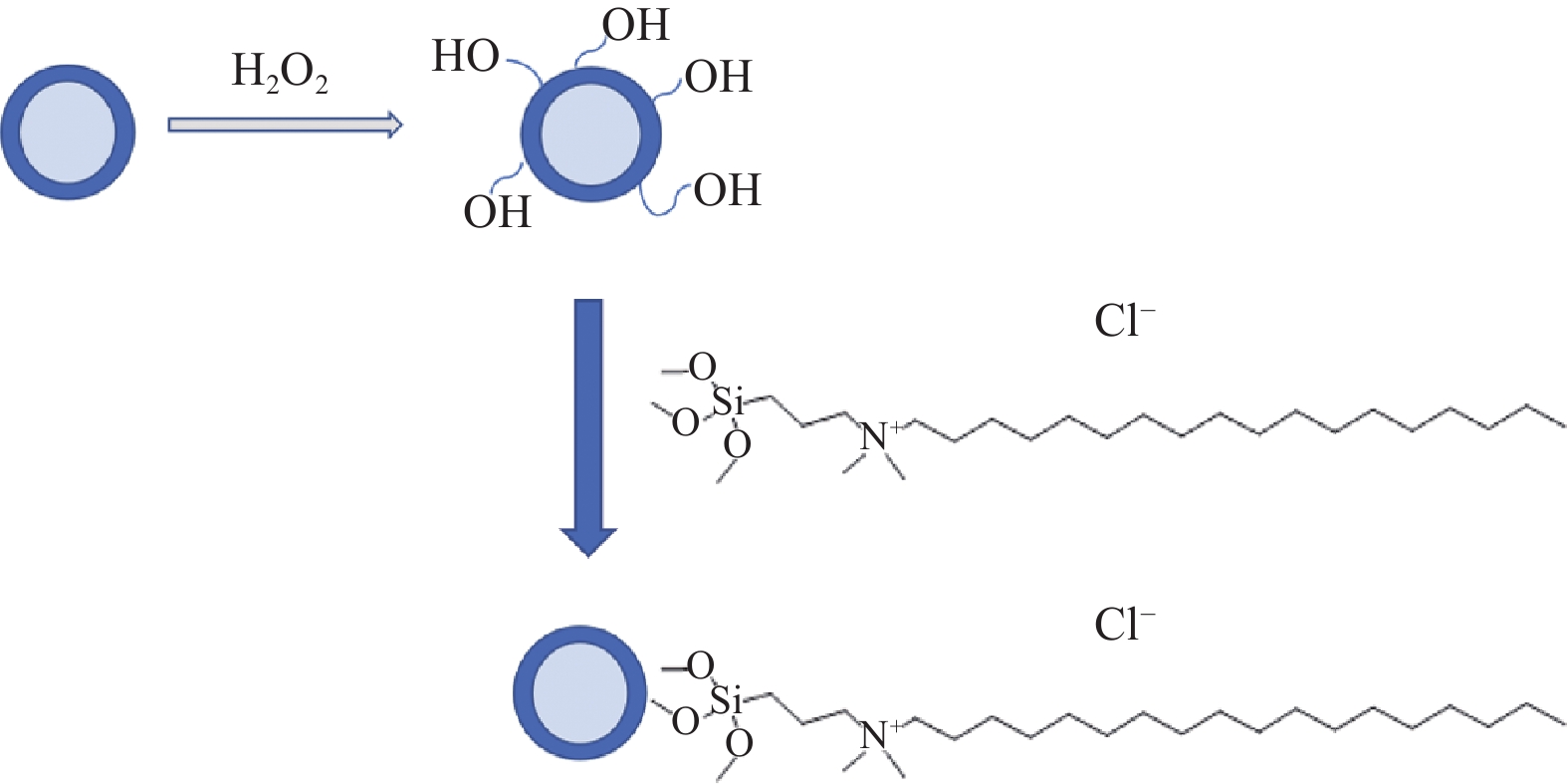
摘要: 为消除金刚线生产过程中分散剂对工艺的负面影响,探索新的金刚线镀镍工艺。首先,使用H2O2对镀镍金刚石颗粒表面进行羟基化;然后,将其同二甲基十八烷基[3-(三甲氧基硅)丙基]氯化铵进行反应,制备表面带正电的金刚石颗粒;最后,对金刚线的制备工艺进行优化。研究结果表明:反应后金刚石颗粒的表面电位从−7.50 mV转变至14.10 mV以上,最高可达30.68 mV。其最佳制备条件为原砂与H2O2的质量比为2∶1,处理时间为1 h;金刚石颗粒与季铵盐的质量比为1∶2,处理时间为4 h。在电镀过程中,经表面化学修饰的金刚石颗粒能够在没有分散剂的条件下,均匀地向阴极迁移,其上砂量增多20%以上。Abstract: A new nickel plating process for diamond wire is explored to eliminate the negative impact of dispersants on the production process of diamond wires. The surface of of Ni-plated diamond particles was first hydroxylated with hydrogen peroxide, and then reacted with dimethyloctadecyl [3-trimethoxysilylpropyl] ammonium chloride to prepare positively charged diamond particles. Finally, the preparing process of diamond wires were optimized. Results show that the surface potential of diamond particles increases from −7.50 mV to 14.10 mV or above after reaction, up to 30.68 mV. The best preparing conditions are as follows: The mass ratio of raw sand to hydrogen peroxide is 2 to 1, with treating time of 1 h; The mass ratio of diamond particles to quaternary ammonium salts is 1 to 2, with treating time of 4 h. In the electroplating process, the diamond particles with chemically modified surface would uniformly migrate towards the cathode without dispersants, the sanding capacity of which could be increased by 20% or more.
-
Key words:
- diamond wire /
- nickel coating /
- chemical modification /
- electroplating
-
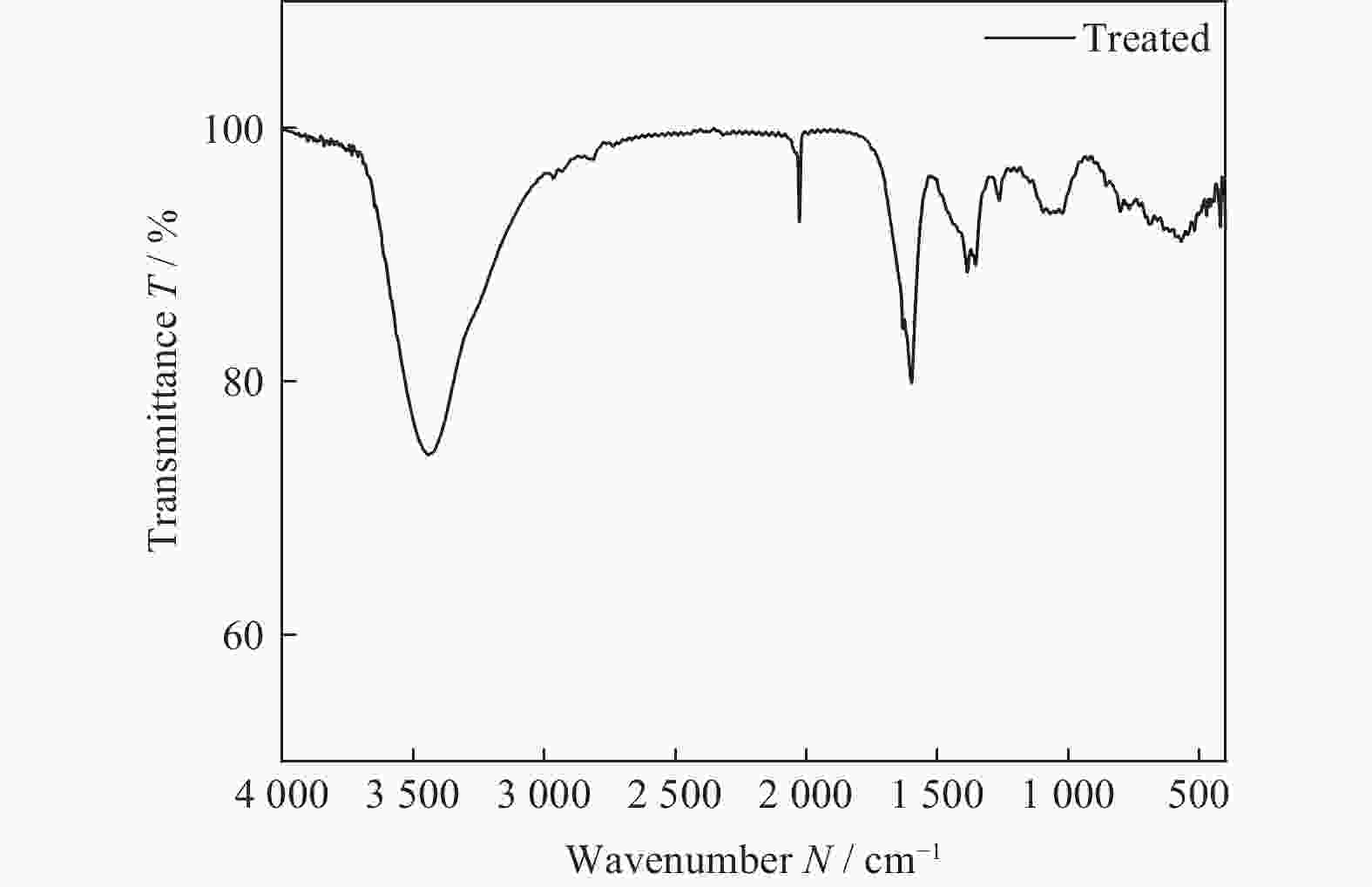
表 1 H2O2处理前后镀镍金刚石颗粒表面元素分布
Table 1. Element distribution on surfaces of Ni-plated diamonds before / after H2O2 treatment
样品 元素 物质的量分数 n / % 质量分数 w / % 原料 O 2.92 0.81 Ni 97.08 99.19 处理后 O 3.37 0.94 Ni 96.63 99.06 表 2 镀镍金刚石粒子表面化学修饰条件优化及对应Zeta电位
Table 2. Surface chemical modification conditions and corresponding Zeta potential of Ni-plated diamond particles
序号 过氧化氢
VOH / mL时间
tOH / h季铵盐
mNH / g时间
tNH / hZeta电位
ζ / mV1 10.0 6 4 8 19.79 2 10.0 3 2 8 14.10 3 10.0 1 2 8 20.05 4 3.0 1 4 8 17.73 5 2.0 1 10 8 20.05 6 1.0 1 2 8 27.17 7 1.0 1 2 4 25.17 8 0.5 1 2 4 30.68 9 0.0 0 1 4 15.80 表 3 公斤级中试颗粒测试条件
Table 3. Test conditions of kilogram scale pilot scale experiment
样品编号 打片时长
tm / min镀液中颗粒
漂浮情况镀液中颗粒
抱团情况3#点位
对比6#点位
对比11 0.5 无 无抱团 多 多 12 1.0 无 无抱团 多 多 13 2.5 无 无抱团 多 多 -
[1] WANG Y, HUANG S, QIAN Z, et al. Modeling and experimental investigation of monocrystalline silicon wafer cut by diamond wire saw [J]. Engineering Fracture Mechanics,2023,278:109029. doi: 10.1016/j.engfracmech.2022.109029 [2] LI A, HU S, ZHOU Y, et al. Recent advances in precision diamond wire sawing monocrystalline silicon [J]. Micromachines,2023,14(8):1512. doi: 10.3390/mi14081512 [3] 李升, 高玉飞, 李新颖. 工艺参数对悬砂法复合电镀制备金刚线的影响 [J]. 电镀与涂饰, 2018, 37(15): 666-670 doi: 10.19289/j.1004-227X.2018.15.002LI Sheng, GAO Yufei, LI Xinying. Effects of process parameters on manufacturing of diamond wire saw by composite electroplating for embedment of abrasive particles [J]. Electroplating and Finishing, 2018, 37(15): 666-670. doi: 10.19289/j.1004-227X.2018.15.002 [4] 金星, 孔剑, 王敬强, 等. 晶体硅金刚线切割废料制备高纯氮化硅 [J]. 应用化学,2018,35(11):1364-1368. doi: 10.11944/j.issn.1000-0518.2018.11.170414JIN Xing, KONG Jian, WANG Jingqiang, et al. Preparation of high-purity silicon nitride from diamond-wire cutting waste [J]. Chinese Journal of Applied Chemistry,2018,35(11):1364-1368. doi: 10.11944/j.issn.1000-0518.2018.11.170414 [5] 陈志坤, 罗涛, 彭必雨, 等. 羟基化改性磷脂的制备及其加脂性能 [J]. 中国皮革,2022,51(1):1-6. doi: 10.13536/j.cnki.issn1001-6813.2022-001-001CHEN Zhikun, LUO Tao, PENG Biyu, et al. Preparation and fatli-quoring properties of hydroxylated phospholipids [J]. China Leather,2022,51(1):1-6. doi: 10.13536/j.cnki.issn1001-6813.2022-001-001 [6] 单国雷, 王龙, 孙元, 等. 镍基单晶高温合金资源中关键金属的浸出行为研究 [J]. 材料导报,2021,35(10):10134-10138. doi: 10.11896/cldb.20040013SHAN Guolei, WANG Long, SUN Yuan, et al. Study on the leaching behavior of key metals in nickel-based single crystal superalloy scraps [J]. Materials Reports,2021,35(10):10134-10138. doi: 10.11896/cldb.20040013 [7] YAO K, DAI B, MAY W P, et al. Hydrophobicity and adhesion of aggregated diamond particles [J]. Physica Status Solidi A,2020,218(5):2000355. doi: 10.1002/pssa.202000355 [8] 王哲, 黄玉东, 岳姗姗, 等. 水溶性聚季铵盐修饰多壁碳纳米管的制备 [J]. 黑龙江大学自然科学学报,2007,24(6):787-790. doi: 10.3969/j.issn.1001-7011.2007.06.020WANG Zhe, HUANG Yudong, YUE Shanshan, et al. Synthesis of water-soluble multi-walled carbon nanotubes covalently bonding polyquaternium salt [J]. Journal of Natural Science of Heilongjiang University,2007,24(6):787-790. doi: 10.3969/j.issn.1001-7011.2007.06.020 [9] 郭慧, 彭学刚, 盛海同, 等. 纳米金刚石的表面修饰及应用 [J]. 化学通报,2014,77(8):778-782. doi: 10.14159/j.cnki.0441-3776.2014.08.018GUO Hui, PENG Xuegang, SHENG Haitong, et al. Surface modification and application of nanodiamond [J]. Chemistry,2014,77(8):778-782. doi: 10.14159/j.cnki.0441-3776.2014.08.018 -



 下载:
下载:





 邮件订阅
邮件订阅 RSS
RSS
